|
Aspergillomarasmine A
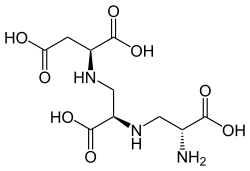
Aspergillomarasmine A is an polyamino acid naturally produced by the mold Aspergillus versicolor. The substance has been reported to inhibit two antibiotic resistance carbapenemase proteins in bacteria, New Delhi metallo-beta-lactamase 1 (NDM-1) and Verona integron-encoded metallo-beta-lactamase (VIM-2), and make those antibiotic-resistant bacteria susceptible to antibiotics.[1] Aspergillomarasmine A is toxic to leaves of barley and other plants, being termed as "Toxin C" when produced by Pyrenophora teres.[2] The molecule is a tetracarboxylic acid with four -COOH groups. One section of the molecule is the amino acid aspartic acid. This has two alanine[contradictory] molecules attached by substituting a hydrogen on the methyl group with a link to the amine group. Aspergillomarasmine B differs in that the last alanine is replaced by glycine. The crystalline substance was first isolated in 1956, but its name was given until 1965.[3] In addition to Aspergillus versicolor, aspergillomarasmine A is also produced by the ascomycete Pyrenophora teres where it acts as a toxin in the barley net-spot blotch disease. In P. teres, a biosynthetic precursor of aspergillomarasmine A, L,L-N-(2-amino-2-carboxyethyl)-aspartic acid has also been isolated and found to contribute to the phytotoxic properties of this microbe.[4] This precursor, aspergillomarasmine A itself, and a lactam form (anhydroaspergillomarasmine A) are together termed the marasmines.[2] Other producers of aspergillomarasmine A include Aspergillus flavus,[3] Aspergillus oryzae,[5] Colletotrichum gloeosporioides, and Fusarium oxysporum.[2] In mice the LD50 toxic dose of aspergillomarasmine A is 159.8 mg/kg.[6] PropertiesAspergillomarasmine A takes the form of colourless crystals. The chemical is insoluble in common organic solvents, but can dissolve in water under either basic or strongly acidic conditions.[3] Anhydroaspergillomarasmine A, a lactam of aspergillomarasmine A, chemically called [1-(2-amino-2carboxyethyl)-6-carboxy-3-carboxymethyl-3-piperazinone], can also be found in Pyrenophora teres. The relative amount of these two toxins is dependent upon the pH of the growth medium, with lower pH favouring the lactam form.[2] The lactam can be hydrolyzed to aspergillomarasmine A by treating it with trifluoroacetic acid.[2] Aspergillomarasmine A functions as a chelating agent, sequestering Fe3+ ions.[7] It can inhibit endothelin converting enzymes even in the live rat, probably by chelating metals required by metalloproteases.[8] When heated, aspergillomarasmine A decomposes between 225° and 236 °C. Hydrolysis produces L-aspartic acid and racemic[why?] 2,3-diaminopropionic acid. Even though the precursor component is chiral, 2,3-diaminopropionic acid easily racemizes in acid.[3] Aspergillomarasmine A has [α]20°D at pH 7 of -48°.[3] With nitrous acid aspergillomarasmine A is deaminated,[clarification needed] and isoserine with aspartic acid is formed.[3] Titration reveals changes in ionisation at pK 3.5 and 4.5 due to carboxylic acid groups, and pK 9.5 and 10 due to amino groups.[3][clarification needed] Treatment with ninhydrin shows a purple colour.[3] References
|
||||||||||||||||||||||||||||||||||
Portal di Ensiklopedia Dunia